




Design
of photocatalysts
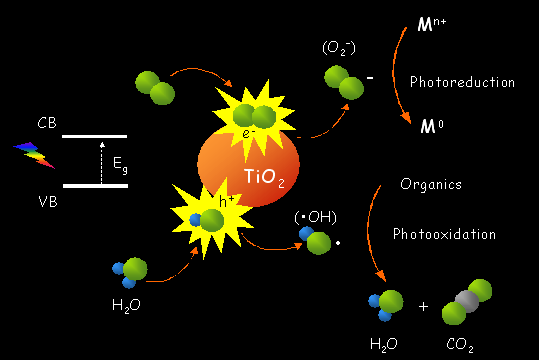
Titanium Dioxide, TiO2, is considered a safe substance
and harmless to human. It is commonly used in paint, printing ink, plastics,
paper, synthetic fibers, rubber, condensers, painting colors and crayons,
ceramics, electronic components along with food and cosmetics. TiO2
can be effectively employed in catalytic reactions such as selective reduction
of NOx to N2, decomposition
of VOCs (including dioxines and chlorinated compounds), hydrogen production
by gas shift production, Fischer-Tropsch synthesis, CO oxidation by O2,
reduction of SO2 to S by CO, and NO2
storage.
We
have focused our attention on the:
> Development of N and B doped TiO2 for dyes
degradation and after suitable metal loading for hydrogen production from
alcoholic solutions;
> Development of Nano Semiconductor Metal Oxides such as ZnO, NiO,
WO3, Bi2O3
and their mixtures in various proportions by sol-gel & co-precipitation
technique for hydrogen production (either by bio-ethanol or water splitting)
as well as environmental applications.

Selected
recent publications
1. V. Gombac, L. Sordelli, T. Montini, J. J. Delgado, A. Adamski, G. Adami,
M. Cargnello, S. Bernal and P. Fornasiero, “CuOx-TiO2
Photocatalysts for H2 Production from Ethanol and
Glycerol Solutions.”, The Journal of Physical Chemistry A 114
(2010), 3916-3925, doi:10.1021/jp907242q.
2. D. Barreca, P. Fornasiero, A. Gasparotto, V. Gombac, C. Maccato, T.
Montini and E. Tondello, “The potential of supported Cu2O
and CuO nanosystems in photocatalytic H2 production.”,
ChemSusChem 2 (2009), 230-233, doi:10.1002/cssc.200900032.